The proposed merger of the National Board of Chiropractic Examiners and Federation of Chiropractic Licensing Boards was approved by NBCE delegates and FCLB members at their respective annual meetings, held jointly in Atlanta, Ga., this year. Per the new bylaws, the new entity takes the NBCE name, with FCLB continuing as a department within NBCE. The federation will continue to enjoy Board of Directors representation on what will be a single, expanded board.
| Digital ExclusiveIs it Osteoporosis of Multiple Myeloma
The films presented below are of an 83-year-old female with complaints of chronic back pain over the past 20 years. She states that she can tolerate a certain degree of back pain, but it has recently increased beyond what she was able to tolerate.
Patients with this degree of osteoporosis are very difficult to evaluate radiographically. A urinalysis and complete blood count (CBC) were performed, and mild anemia and hypercalcemia were the results. These two findings indicate some systemic disorder. Where would you go from here?
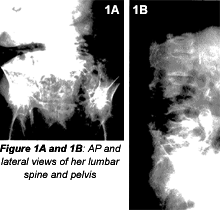
From this information, I would probably order a bone scan. A clinician with expertise in internal medicine would probably order further blood tests, most likely including a serum protein electrophoresis (SPEP), because of the decreased number of red blood cells found in the CBC. I cannot tell you which to do first, because it depends on the patient's symptoms and clinical history. In this case, I was concerned about possible metastatic lesions from an unknown primary and possible multiple myeloma. Those are the two disorders that I learned were very important to rule out, especially in patients over 70, before less aggressive disorders should be considered. My other concern was possible pathologic fracture due to the severe osteoporosis.

The bone scan demonstrates a slightly asymmetric uptake of the radionuclide in the sacroiliac region, but it is not particularly helpful when attempting to determine a differential diagnosis. By now, most of you know what the diagnosis probably should be in this case. A CT of the lumbar spine and pelvis was performed along with SPEP. A lytic lesion was found in the sacrum, and the SPEP revealed an M-protein spike. Bone marrow aspiration confirmed the diagnosis of multiple myeloma.
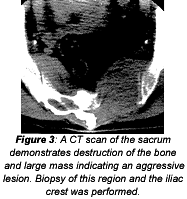
Invasive Versus Noninvasive Tests
Before I continue regarding multiple myeloma, I would like to suggest repeating any noninvasive tests before recommending a bone biopsy or bone marrow aspiration. It is not an easy test for the patient, and lab errors do occur. It is much easier to draw another vial of blood just to confirm a finding before undergoing any invasive procedure. As a case-in-point, approximately nine years ago my father had a SPEP study performed; the results were equivocal. The internist recommended a bone marrow aspiration to rule out multiple myeloma. We decided not to do the bone marrow aspiration until another SPEP was done. It came back normal! Thank goodness there was no need for any bone aspiration.
Though the bone marrow aspiration and bone biopsy sound rather minor, removing a sample of marrow or bone is not without discomfort or possible infection. The so-called "needle" used is fairly large, with a hollow core and a spade-shaped tip, which is pushed through the cortex of the bone into the marrow cavity with an advancing clockwise-counterclockwise motion. The specimen needs to be approximately 1.5 to 2.0 cm in length.
Needless to say, these are not friendly-looking devices. I think I have made my point, with puns intended!
Multiple Myelomas
Multiple myeloma is the second most prevalent blood cancer and represents approximately one percent of all cancers, and two percent of all cancer deaths. The characteristic age of incidence is 65 to70, and statistics seem to indicate that the incidence and younger age of onset are increasing.
Multiple myeloma is more common in males. African Americans and Native Pacific Islanders have the highest incidence of this disease; Asians have the lowest.
The cause of multiple myeloma is not known; however, there seems to be a possible association between myeloma and a decrease in the immune system. Genetic factors, exposure to certain chemicals, radiation and a possible virus are all being evaluated as possible causes.
All the usual diagnostic studies that you learned in school are still used to determine the diagnosis of multiple myeloma: The large M-protein spike from electrophoresis of the serum and urine is still an indicator. Electrophoresis used on urine samples can also detect Bence Jones proteins. The multiple lytic lesions in the bone are also a classic finding.
If a positive diagnosis is made, several further tests are done to assess the extent of the disease, to plan treatment, and to establish a baseline against which to measure the effectiveness of treatment.
Increased blood urea nitrogen (BUN) and creatinine levels indicate decreased kidney function.
Bone marrow aspiration or biopsy may determine the percentage of plasma cells in the marrow. Multiple myeloma is probable if 10 percent of the cells in the sample are plasma cells.
Clinical stages of myeloma are based on several criteria: the number of lytic bone lesions; hemoglobin value; serum calcium; and kidney function. I am not an expert on this topic, but proper staging helps determine a treatment plan and prognosis. The most accepted staging for myeloma is the Durie-Salmon system, which can be found throughout the literature that discusses myeloma.
There are also two other categories that are not included in the staging system. The first is "monoclonal gammopathy of undetermined significance (MGUS)." As is implied by its name, we don't know exactly what it is. It is considered a precancerous asymptomatic condition that demonstrates slightly increased plasma cells in the bone marrow, with slightly elevated M-protein. This may be significant because 30 percent of patients with this condition will develop B-cell malignancy within 15 years. The condition is not treated at this point, but the patient is evaluated regularly to determine if there is any progression of the disease.
The other classification is "smoldering myeloma," in which the patient is asymptomatic with increased plasma cells in the bone marrow, elevated M-protein, and a slowly progressing or stable disease. Again, the patient is evaluated regularly to determine if there is any change in the condition. Patients with stage I of the disease are monitored every three months; treatment ensues only if there is progression of the disease. Patients with Stage II and III have what is called "active myeloma" and are treated immediately.
The standard treatment for multiple myeloma includes chemotherapy; chemotherapy plus stem cell transplantation; and radiation therapy. Treatments are emerging that offer more options. Research in this area is constantly improving patients' prognoses.
In a case I recently reviewed, the patient's films did not usually demonstrate the classic findings that we learned in school. By the time there are multiple lesions in the bone, the patient is in stage II or III. A patient's best chance for survival is long before there are multiple "punched-out" lytic lesions in the bone. In my experience, the cardinal sign that I have seen is simply marked osteopenia in a male patient over 70. The only other sign was a rib fracture with no history of trauma.
Remember that the bone destruction is caused by two separate events. The first is rapid growth of myeloma cells that push aside normal bone-forming cells, weakening the bone. Second, the myeloma cells seem to activate substances such as osteoprotegerin ligand and osteoproterin, which are involved in the regulation of bone resorption, causing an imbalance in the breakdown, repair and growth of bone. The osteoclasts are increased, accelerating the breakdown of bone without the balance of repair and growth of bone. The bone changes are secondary signs of this disease - a hematologic cancer of the plasma cells.
Deborah Pate,DC,DACBR
San Diego, California
patedacbr@cox.net



